Acute uncomplicated urinary tract infections (UTI) are one of the most-common outpatient infections, experienced by 50% to 60% of adult women at some point in their lifetime. Infection of the urinary tract can occur when bacteria enters the urethra from bacterial sources such as the skin or rectum. Associated UTI societal costs — such as for healthcare as well as work absences — are projected to be $3.5 billion per year in the United States.
A serious complication of UTIs is pyelonephritis, where the infection progresses from the bladder and infects the kidneys, which can lead to hospitalization and permanent kidney damage.
Risk factors for acute uncomplicated UTI
Certain people have a higher risk of UTI:
- Females (urethras are shorter and close to rectum allowing bacteria to enter more easily)
- Previous UTI
- Sexual activity
- Pregnancy
- Urinary tract structural abnormalities
- Changes in vaginal bacteria or flora
- Poor hygiene
Symptoms
- Pain and burning when urinating
- Frequent urination
- Feeling the need to urinate when bladder is empty
- Voiding small volumes of urine
- Blood in urine
- Groin or lower abdominal pressure or cramping
Consult the IDSA uncomplicated UTI clinical guidelines
UTIs are classified as either complicated or uncomplicated. Uncomplicated UTIs are those present in immunocompetent, nonpregnant, premenopausal persons with no comorbidities or known urologic abnormalities.
The Infectious Diseases Society of America (IDSA) clinical practice guidelines for acute uncomplicated cystitis in women were updated in 2010, stemming from increasing UTI-causing uropathogen antimicrobial resistance and variances in prescribing practices despite existing guidelines. The American Journal of Obstetrics and Gynecology reported that 58% of prescribing practices for UTI were in concordance with IDSA guidelines.
Treatment is based on therapy risk assessment. The choice of agent is individualized, taking into consideration adverse effect risk, allergy, effectiveness and resistance rates.
Consider drug reactions that could increase antimicrobial resistance in uncomplicated UTI
Since UTIs are very common, maintaining antimicrobial effectiveness and reducing collateral damage is important. Collateral damage refers to adverse ecological effects of antimicrobial therapy that includes infections with multi-drug resistant organisms or treatment failure and the selection of drug-resistant organisms. Regional antimicrobial resistance rates are a notable consideration in the selection of drug therapy, and resistance is specifically addressed in the revised clinical guidelines.
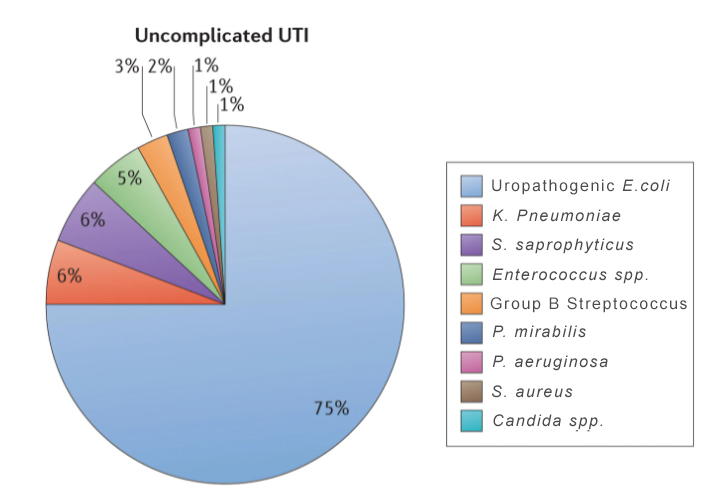
Escherichia coli is the predominant causative pathogen in uncomplicated cystitis, accounting for approximately 75% of UTIs. Local E. coli antimicrobial susceptibility should be considered when selecting antibiotic treatment and prior to initiating empirical therapy. Increasing trends have been observed for empirical UTI treatment with antibiotics in the absence of performing or obtaining results from urine cultures or susceptibility testing. Depending on the prescribed medications, this practice can lead to failed therapy, resistance or collateral damage.
Nitrofurantoin monohydrate, Trimethoprim-sulfamethoxazole, and fosfomycin trometamol are first-line agents when taking into consideration efficacy, resistance and propensity for collateral damage. However, Trimethoprim-sulfamethoxazole exhibits some patterns of local resistance, and should not be used first line if the resistance prevalence exceeds 20% in the prescribing region. Fosfomycin has minimal resistance and low propensity for collateral damage, but has reports of lower efficacy than other short-course UTI drug regimens.
Beta-lactam antibiotics exhibit E. Coli resistance greater than 20%. They are not first-tier agents if other agents can be used. Empirical therapy with amoxicillin or ampicillin should not be initiated due to high antimicrobial resistance and poor empirical efficacy.
Fluoroquinolones are very efficacious, but have a higher propensity for collateral damage. Resistance is below 10% in North America and Europe, but resistance has been increasing. To preserve their effectiveness for other medical uses, fluoroquinolones are not a first-line treatment option for UTI if other agents are suitable based on treatment risk.
Treatment: Acute uncomplicated cystitis in women
UTI drug regimens
First-line
- Nitrofurantoin (macrocrystals) 100 mg twice per day for five days
- Trimethoprim/sulfamethoxazole (Bactrim, Septra) 160/800 mg twice per day for three days if local resistance does not exceed 20%
- Fosfomycin (Monurol) 3g as a single dose
Alternative antimicrobials
B-Lactam agents
- Amoxicillin/clavulanate (Augmentin) 500/125mg twice per day for 5 to 7 days
- Cefdinir (Omnicef) 300mg twice per day for 5 to 7 days
- Cefodroxil 500 mg twice per day for 5 to 7 days
- Cefpodoxime proxetil 100 mg twice per day for 5 to 7 days
Fluoroquinolones
- Ciprofloxacin (Cipro) 250 mg twice per day for three days
- Ciprofloxacin, extended release (Cipro XR) 500 mg per day for three days
- Levofloxacin (Levaquin) 250 mg per day for three days
- Ofloxacin 200 mg per day for three days or 400-mg single dose
Pharmacists and uncomplicated UTIs
Pharmacists — with their deep knowledgeable of clinical practice guidelines, treatment options, prevention strategies, supportive care and self-care — serve as a resource to both physicians and patients through treatment recommendations and education. Recognizing the significance of antimicrobial resistance and collateral damage, combined with their specialized medication knowledge, pharmacists can coordinate with physicians to improve UTI patient outcomes while preserving antimicrobial effectiveness. For more information on managing UTIs, check out our blog.
Resources
Diagnosis and Treatment of Acute Uncomplicated Cystitis, American Family Physician
International Clinical Practice Guidelines for the Treatment of Acute Uncomplicated Cystitis and Pyelonephritis in Women. Oxford Academic
Preventing and Managing Urinary Tract Infections: Enhancing the Role of Community Pharmacists — A Mixed Methods Study. Antibiotics